Last Updated on October 9, 2024 by The Health Master
Drug recall
High Blood Pressure (High BP) drug Recall, also known as hypertension drug recall, is a critical public health update for individuals taking medication to manage their blood pressure.
This article details a recent drug recall of a specific blood pressure medication by Glenmark Pharmaceuticals due to concerns about its effectiveness.
Glenmark Recalls Diltiazem Hcl Capsules
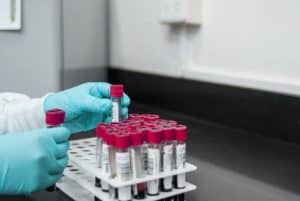
According to a recent Enforcement Report by the U.S. Food and Drug Administration (USFDA), Glenmark Pharmaceuticals, a major pharmaceutical company, is recalling 6,528 bottles of their Diltiazem Hydrochloride extended-release capsules.
Diltiazem Hydrochloride is a medication prescribed to treat high blood pressure (high BP hypertension).
Reason for the Drug Recall: Failed Dissolution Specifications
The USFDA cites “failed dissolution specifications” as the reason behind the drug recall. Dissolution refers to the process by which a medication breaks down and releases its active ingredients into the body.
Medications are expected to maintain consistent dissolution rates throughout their shelf life.
In this case, a test conducted at the 12th month mark of a long-term stability study revealed that the Diltiazem Hydrochloride capsules did not meet the required dissolution specifications.
Potential Impact of the Drug Recall
The USFDA has classified this drug recall as a Class II recall.
This classification signifies that the use of the affected medication may cause temporary or reversible health issues.
There’s a remote possibility of serious health consequences.
However, it’s crucial for patients taking the recalled medication to consult their healthcare provider to discuss alternative treatment options to ensure uninterrupted blood pressure management.
What to Do if You Have the Recalled Medication
- Identify the Batch Number: The drug recall affects only specific batches of Diltiazem Hydrochloride capsules. Check the medication bottle for the batch number and expiration date.
- Stop Taking the Medication: If you have a bottle included in the drug recall, discontinue taking the medication immediately.
- Contact Your Doctor: Schedule an appointment with your doctor to discuss alternative treatment options for managing your blood pressure.
- Return the Medication: Follow Glenmark Pharmaceuticals’ instructions for returning the recalled medication.
Glenmark Initiates Nationwide Drug Recall
Glenmark Pharmaceuticals initiated a nationwide Class II recall of the affected Diltiazem Hydrochloride capsules across the US on March 26, 2024.
Patients with any questions or concerns regarding the drug recall can contact Glenmark Pharmaceuticals.
Maintaining Blood Pressure Control is Essential
High blood pressure (high BP) is a significant health concern that can lead to serious health complications if left untreated.
It’s vital for individuals taking medication for hypertension to ensure they are receiving effective treatment.
If you have any concerns about your blood pressure medication or have been taking the recalled Diltiazem Hydrochloride capsules, consult your healthcare provider for guidance and alternative treatment options.
Disclaimer: This article contains information derived from the source mentioned below. Our team utilized an AI language model to rewrite and present the news or article in a unique format.
Drug recall: Drugs recalled by these Indian Pharma Companies
Drug recall: Mesalamine capsules recalled Due to this reason
Drug recall: Dextroamphetamine sulfate tablets recalled Due to this reason
Drug recall: Various Eye drops recalled due to this reason
Drug recall: Dr Reddy’s recalls over 8,000 bottles of generic drug due to packaging error
Drug recall: Benzocaine Topical Anesthetic Spray recalled Due to this reason
CDSCO approval granted for this game changer Eye Drop
FSSAI: Food Labelling and Display – Chapter-4
Rs 36 Lakh Worth of Pharma Raw Material stolen
CDSCO releases draft Guidelines for Clinical Trial in India
DCC Approves Ban on Harmful Antibiotics in Food Products
DCC: Sub-committee to look into one-time NOC for exports
CDSCO: New draft Guidelines for Pharmaceutical Distribution in India
Codeine Phosphate and its Regulations in India
USFDA approval granted for Fluoxetine Tablets
Belarus Seeks 84 Medicines: A Lucrative Opportunity for Indian Pharma