A new study by scientists from India, USA, and UK has presented clinical evidence for an innovative test that can detect clusters of cancer cells in the blood of asymptomatic individuals as a non-invasive screening and diagnostic method.
The test makes cancer screening easier, efficient and affordable and can potentially be a breakthrough in cancer detection and diagnosis.
The test will soon be available commercially.
Early detection of cancer is crucial but challenging because of the lack of efficient and reliable screening methods.
Most of the commercially available cancer-screening tests are invasive and expensive.
Also, currently available cancer screening techniques such as mammograms and low-dose CT scans (LDCT) carry radiation risks, colonoscopies are invasive, blood based markers are non-specific and tissue biopsies for diagnosis have the same risks as general surgical procedures.
Also read: PGI gets 3,000 cancer cases annually
Commenting on the study, principal author Dr. Dadasaheb Akolkar, who is the research director at Datar Cancer Genetics said, “This is the first study of its kind to investigate the prevalence of circulating tumor emboli or Circulating Ensembles of Tumor Associated Cells (C-ETACs) in over 16,000 participants to establish the definitive new systemic hallmark of cancer.
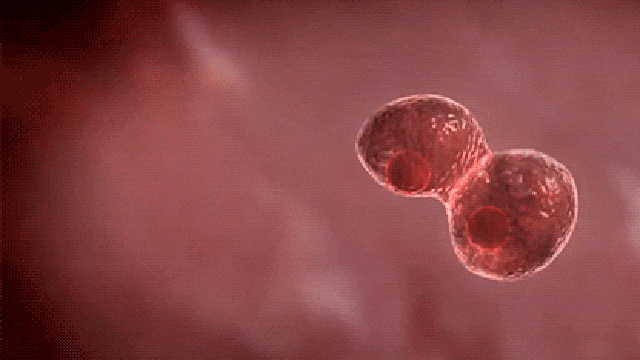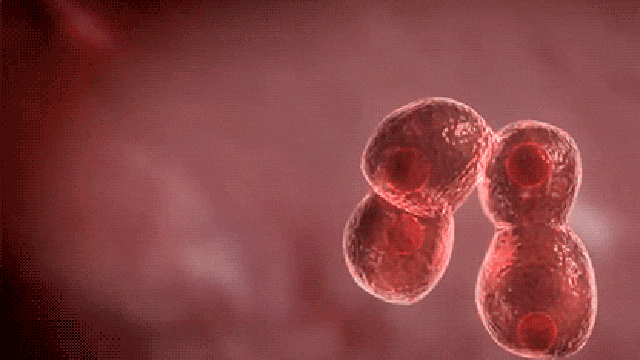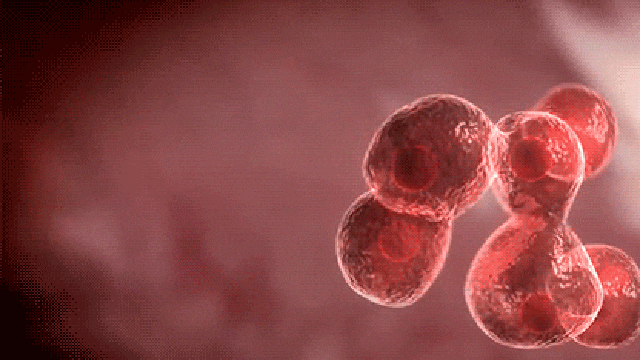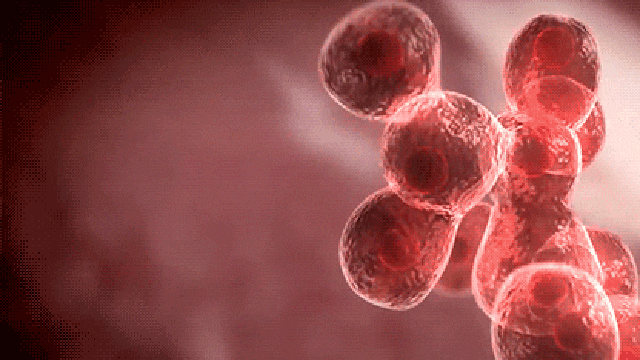
The technique we have used is a breakthrough innovation.
When clusters of cells break off from an early stage tumor and enter the bloodstream, we can efficiently and accurately isolate a few hundred malignant cells from more than 100 million cells, using just 10 ml of blood.
While almost all cancer samples had these cell clusters, they were seen in very few of the samples which were apparently without cancer.”
Speaking on the breakthrough study and technique, Rajan Datar, chairman and managing director, Datar Cancer Genetics said, “Cancer is rapidly becoming a civilizational challenge. Importantly, cancer deaths are mainly because of late detection.
Also read: USFDA approves Merck’s Keytruda for bladder cancer
We believe that this innovative blood-based test is a breakthrough in cancer screening and will impact outcomes through easy, patient-friendly detection and diagnosis in apparently healthy people who may have a silent malignancy in their bodies.
It has the potential to eliminate the need for invasive biopsies and the risks associated with it.
In the near future, a simple, inexpensive blood test that could be all that is required to reliably detect and diagnose cancer, even before any symptoms are seen.”
The study involved 16,134 participants, including 5,509 patients with cancer (TrueBlood study) and 10,625 individuals with no symptoms (RESOLUTE study) and the test has shown an accuracy of more than 94%.
Also read: USFDA nod to Lynparza to treat pancreatic cancer
The C-ETACs were seen in 89.8% of cancer cases and in only 3% of apparently healthy, asymptomatic individuals who had no abnormal findings in presently used screening tests.
The study was the largest of its kind in the world.
Datar Cancer Genetics has also presented further data at several leading international conferences including AACR, ASCO and ESMO
Datar Cancer Genetics is a leading molecular oncology facility with a fully equipped laboratory, integrated process platforms, in-house bioinformatics team, and a huge genetic database for precise and updated reporting.
Also read: Health Insurance Cos. introduce Cancer Protect Cover starting Rs 125


