Last Updated on January 14, 2024 by The Health Master
21 hospitals shortlisted for plasma trials: ICMR
NEW DELHI: The Indian Council of Medical Research (ICMR) has finalised 21 hospitals to initiate clinical trials for convalescent plasma therapy, which uses antibodies from the blood of cured patients, to treat critically ill Covid-19 patients. The most number of hospitals chosen are in Maharashtra.
ICMR has finalised as under:
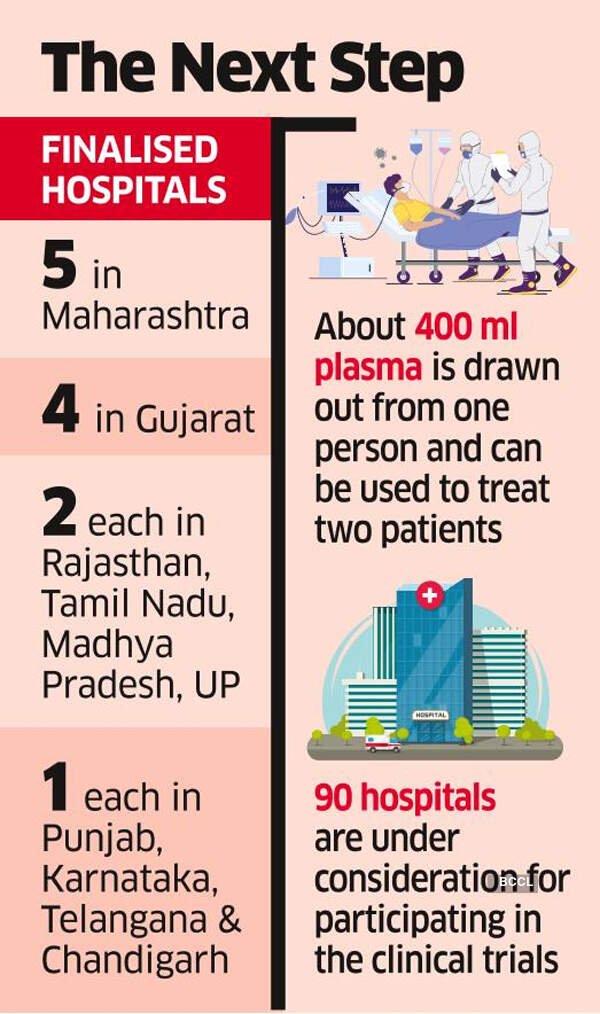
Five hospitals in Maharashtra,
four in Gujarat, two each in Rajasthan, Tamil Nadu, Madhya Pradesh and Uttar Pradesh, and
one each in Punjab, Karnataka, Telangana and Chandigarh to initiate the clinical trials.
To download the list of hospitals, click here
Plasma therapy
The plasma therapy involves drawing out whole blood from a patient, who has recovered from Covid-19, and separating the plasma. About 400 ml plasma is drawn out from one person, which can be used to treat two patients.
Also read: Dr Reddy’s launches generic hemophilia inj.
Maharashtra Hospitals
Of the five hospitals in Maharashtra, two are in Pune, one in Mumbai, one in Nagpur and one in Kolhapur. Sir H N Reliance Foundation Hospital and Research Centre of Mumbai has been chosen for the trials. The multi-centre trials would be conducted on 452 patients across centres.
ICMR’s project is called PLACID — Phase-II Open Label Randomised Controlled Trial to Assess the Safety and Efficacy of Convalescent Plasma to Limit Covid-19 Associated Complications in Moderate Disease. The study received approval from the Covid-19 National Ethics Committee (CONEC) last week.
90 hospitals are in queue
About 90 hospitals, including seven in Delhi, are under consideration for participating in the clinical trials. Hospitals in the capital which are awaiting approval include AIIMS, Indraprastha Apollo, Batra Hospital and Medical Research Centre, Lady Hardinge Medical College, RML Hospital, Max Hospital-Saket and Indian Red Cross Society.
On April 29, the health ministry had termed as “illegal” any use of the therapy beyond clinical trials. “The therapy can create life threatening conditions,” the ministry had said. Plasma therapy was used at the time of SARS outbreak on critically ill patients. The therapy works as a passive immunity system.
The Health Master is now on Telegram. For latest update on health and Pharmaceuticals, subscribe to The Health Master on Telegram.


